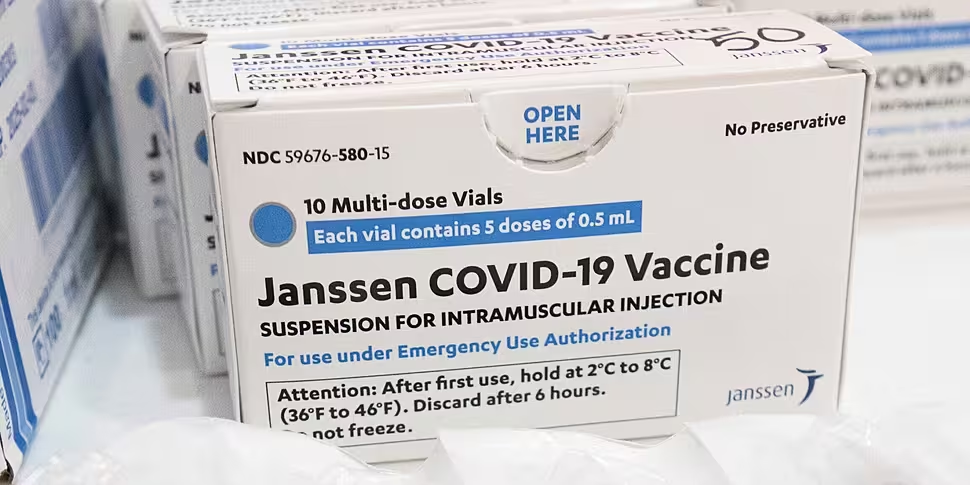
The European Medicines Agency (EMA) says it has found a 'possible link to very rare cases of unusual blood clots' in people with low blood platelets who receive the Johnson & Johnson vaccine.
But it says the overall benefit-risk 'remains positive'
The EMA's safety committee says a warning about unusual blood clots with low blood platelets "should be added to the product information".
It also says these events should be listed as very rare side effects of the vaccine.
The committee says it took all currently available evidence into consideration - including eight reports from the United States of serious cases of unusual blood clots associated with low levels of blood platelets.
One of these had a fatal outcome, it adds.
All cases were in people under 60 years of age within three weeks after vaccination, the majority of which were women.
It adds that specific risk factors have not been confirmed.
However it suggested one plausible explanation for the combination of blood clots and low blood platelets is "an immune response, leading to a condition similar to one seen sometimes in patients treated with heparin called heparin induced thrombocytopenia, HIT".
As April 13th, over seven million people had received the Johnson & Johnson - or Janssen - vaccine in the United States.
Earlier this month, the company delayed the rollout of its COVID-19 vaccine in Europe.
The company said it made the decision 'proactively' after US authorities recommended pausing the rollout there.
The first doses of the one-shot vaccine were due to arrive in Ireland that same week.